NONRENEWABLE
ENERGY
SOURCES AND HOW THEY ARE USED
Unit Overview
In this unit, nonrenewable
sources of energy will be defined and investigated. Their use in our society to provide for our
basic needs of electricity, transportation, and heating will be discussed. Energy transformations will be noted, as well
as the efficiency of those transformations.
We have studied the seven
forms of energy. Some were classified as
potential – nuclear, chemical, and mechanical energy (due to its position or
condition). Others were classified as
kinetic energy – active mechanical energy, sound or acoustic energy, thermal
energy, electrical energy, and radiant energy.
Now we will discuss where we get these forms of energy; that is, what
are the sources of our energy. In this unit we will study the nonrenewable
sources, those that cannot be replaced.
As we discover the various uses for these sources, we will follow their
transformations through the various energy forms.
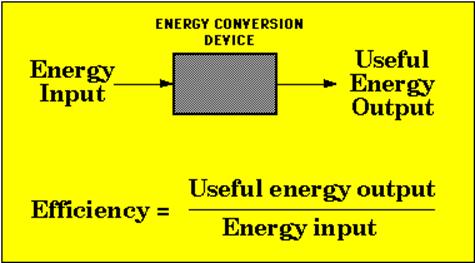
Efficiency
If you remember from our
earlier discussions of the energy pyramids, we noticed that energy
transformations always involve some loss of useable energy to what we term
waste heat. It is called waste because
it serves no useful purpose. This is
true in any conversion of energy from one form to another. A way to determine just how much useful
energy is derived from the conversion is to determine efficiency. Mathematically, efficiency can be calculated
by a fairly simple formula as seen in the diagram below. The total amount of energy put into the
process which is measured in an energy unit such as joules is placed in the
denominator of a fraction. The total
amount of useful energy produced by the process, measured in the same energy
units as the input, is placed in the numerator.
The larger the numerator, the greater the efficiency of the
process. To express the efficiency in a
percent we simply multiply by 100. So if
a process is 50% efficient that means that only half of the original input
energy is being used.
Petroleum

An oil rig in the ocean.
The greatest source of energy
in the United States is petroleum. This
fossil fuel is found deep in the earth with the average oil well being 6000
feet below the surface. Because fossil
fuels were formed over many, many years, perhaps millions of years, these
cannot quickly be replaced. Over 38%
of our energy in the United States comes from petroleum, but we provide only a
little over 16% of this from our national resources. The rest is imported from other
countries. We get most of our oil from
Canada, Venezuela, Saudi Arabia, and Mexico.
Texas produces more oil than any other state. The United States uses more petroleum than
any other country in the world, with most of it (over 2/3) being used as a fuel
for transportation. Other uses are for
industry as a source of heat but also as raw material for making fertilizers,
plastics, medicines, and other products.
Only 6% of the petroleum is used for heating in homes and businesses and
1% for the production of electricity.
But even before it can be used in any of these applications, it must
first be refined. Nearly 30% of undiscovered reserves are thought to be
offshore.
|
|
|
|
||
|
Pumping crude oil to the surface (left) |
Crude oil, petroleum as it comes from the
ground.(right) |
||
Petroleum is known as crude
oil when it comes from the ground. It is
a mixture of many different hydrocarbons, chemicals composed of carbon and
hydrogen. It can vary in color from near
colorless to black, and it can be as thin as gasoline or as thick as asphalt
due to the amounts of the various hydrocarbons that it contains. Refining consists of various processes in
which the mixtures are separated into smaller groups and cleaned of contaminants. This requires heat, so about 9% of the
petroleum is used in the refining process alone.
Once it has been separated
into various components, such as gasoline, heating oil, diesel fuels, propane,
jet fuel, asphalt, and others, it can be used.

A petroleum refining facility
What form of energy is found
in petroleum? If you said chemical, you
are right. The chemical energy was stored
there by the plants that once lived and carried on photosynthesis. It is thought that dead sea animals and
plants are the once-living matter that makes up petroleum. Of course, the animals got their food either
directly or indirectly from the plants, so the initial source of the energy was
radiant energy from the sun. As you
recall, plants must have sunlight in order for photosynthesis to occur. When petroleum is used as gasoline in our
cars, the conversion of energy continues.
Inside the engine, the gasoline is burned, being ignited by the spark
plugs. As in all burning, heat and light
are produced, but that is not the end of the story. The rapid burning inside the cylinders of the
engine causes a rapid expansion of the hot gases which pushes the piston
upward. This motion through a system of
gears is transferred to the wheels to make the car move. So we have kinetic mechanical energy in the
end plus lots of waste heat. That is why
we need a radiator and/or air to cool the engine so it does not overheat. It is wasted heat because it serves no useful
purpose except for the occasional use of this heat to warm the inside of the
car during the colder months of the year.
This wasted heat escapes from the radiator or out the exhaust pipe and
goes into the atmosphere. Because so
much energy is wasted, such as this heat, most vehicles are not very
efficient. Only about 25% of the energy
in the gasoline is actually used to move the car, and 75% is wasted as
heat. That makes the efficiency of the average
automobile only 25%. To improve this
efficiency, we often look for ways to get more miles to the gallon of
gasoline. Manufacturers of automobiles
have made some strides in this area by changing the engine, the design of the
car, and the materials used in its construction.
Petroleum can also be used
for heating. Fuel oil can be used for
heating in manufacturing and in our homes.
Here the conversion is from radiant energy from the sun to the stored
chemical energy due to photosynthesis to heat and light as it is being burned
just as it was in the automobile. The
heat can be used to heat living areas or to provide heat for some manufacturing
process. The efficiency here is much better, about 65%. A small amount of petroleum is used in
electric power-generating plants. We
will discuss the energy conversions that occur there later in this unit.
Coal

Strip or surface mining of coal
Like petroleum, coal takes many
millions of years to form so it is known as a nonrenewable energy source. It is thought to have been formed in what
were formerly swampy areas. As the dead
plants and animals accumulated on the bottom, they were packed together by
layers of dirt and water and subjected to heat and pressure. Over time oxygen was released leaving a rich
energy source of hydrocarbons.
Hydrocarbons are compounds consisting of only hydrogen and carbon. This hydrocarbon is coal and exists in seams
within the earth anywhere from a fraction of an inch to hundreds of feet. The Pittsburgh seam which is seven foot thick
may represent 2,000 years of rapid plant growth. So you can see just how much energy is packed
into coal. The United States is the
world leader in known coal reserves and if used at today’s rate, we have enough
to last 300 years. The state with the most coal deposits is Montana with
Illinois, Wyoming, Kentucky, and West Virginia also having significant coal
deposits. In the West, the federal
government owns 60% of the coal with indirect control over another 20%. Western coal tends to be lower in sulfur than
eastern coal and therefore creates less air pollutants.
|
|
|
||
|
Long wall mining deep underground |
A continuous mining machine used in deep mines |
|
Coal is an important energy
source for the United States as it supplies 23%, almost one-fourth, of our
energy needs. Most of its use (90%) is
in the production of electricity. In fact,
the Sammis Power Plant at Stratton, Ohio, and the Cardinal Power Plant at
Brilliant, Ohio, are both coal-fired power plants. Another important use is in the production of
coke to use in the steel-making plants.
There is a coke producing plant in Follansbee, West Virginia. Coke is made by driving impurities from the
coal using heat without the presence of oxygen.
This coke is an excellent fuel for smelting or purifying metals, such as
iron and copper. Whether coal is used
directly as a fuel, as in electric-generating plants, or indirectly, as in the
smelting of metals, it is always used as a heat source. To trace the energy transformations that
occur, we would start with radiant energy from the sun to stored chemical
energy in the fuel to heat when the fuel is burned.
Although both petroleum and
coal are good sources of energy, neither is totally free of contaminants which
create air pollution when they are burned.
They also release carbon dioxide into the atmosphere which, as you will
recall, is one of the gases responsible for global warming. These represent problems that challenge
us. Solutions have been found for
controlling some of the pollutants.
Scrubbers use water and limestone to remove sulfur from the
exhaust. Electrostatic precipitators
remove fly ash, a fluffy, by-product particle produced during the burning of
coal. This can be used as a
road-building material, as a cement additive, and as pellets for oyster beds.
Natural Gas
Another fossil fuel that is
in gaseous form is natural gas. Although
there are renewable sources of this gas, most of it comes from deep underground
trapped in porous rock by itself or with petroleum or coal deposits. Once it is brought to the surface, it is
cleaned of impurities and separated into its various components. Most of natural gas is methane but it does
contain some propane and butane, other hydrocarbon gases. The top five natural gas producing states in
order are Texas, Louisiana, Oklahoma, New Mexico, and Wyoming. Natural gas supplies 22% of out energy needs
or over one-fifth of our needs. About
25% of the world-wide production of natural gas occurs in the United
States.
Natural gas is a very clean
burning fuel with water vapor and carbon dioxide as the only products of
combustion, which makes it a good choice for fuel. The biggest user of natural gas is industry
where it is used as a fuel to produce heat but it is also used as an ingredient
in fertilizers, photographic film, ink, glue, paint, plastics, laundry
detergent, synthetic rubber, insect repellents, and synthetic fibers like
nylon. The second largest use of natural
gas is for home heating. Over half of
the houses in the U.S. have natural gas furnaces. The efficiency of natural gas is fairly high
at 85%. Next to coal and uranium,
natural gas is the third largest producer of electricity in this country. It produces electricity more efficiently than
does coal with much less concern for pollutants.

Natural Gas, a Clean Burning Fuel

Natural gas is transported
through underground pipelines throughout the U.S

Some of the many uses of natural
gas
The supplies of natural gas
in this country are not unlimited. With
today’s known sources and using at today’s rate, we have only a 50 year supply
of natural gas. Scientists feel that
there are enough reserves, areas thought to contain natural gas but not yet
tapped, to last 200 years. However,
getting to these reserves would require extended efforts and would thus raise
the price of natural gas considerably.
Propane

Another fossil fuel, propane,
is taken from natural gas and petroleum deposits. At normal temperatures and pressures, it is a
gas, but it can be easily liquefied or changed into a liquid by placing it
under pressure. If you remember from our
discussion of the states of matter, the particles in a liquid are much closer
together than those of a gas. For this
reason, liquefied propane takes up much less space than gaseous propane. You have probably seen gas grills with the
tanks of propane attached, or perhaps you know someone who heats and cooks with
propane. A large tank of propane is
somewhere nearby to supply these needs.
In both cases, the tanks are filled with liquefied propane which turns
into a gas when released from the pressure inside the tank. Because it can be easily liquefied and placed
in tanks, it is more easily transported than some other fuels. People who live in rural areas may not have
access to natural gas pipelines and instead can use propane as a fuel if they
choose. Still propane serves only about
1.8% of our energy needs which is quite low.
|
|
|
|
Left: a
forklift powered by propane has few emissions and is safe to operate inside. Right: A truck fueled by propane is clean-burning
and has less engine wear. |
|
The
largest use for propane is in industry where it is used for cutting torches,
soldering, vulcanizing (a process to make rubber suitable to be made into
tires), portable heaters for construction sites, keeping asphalt at the proper
consistency for highway work, and to power fork-lifts inside of
warehouses. About one-third of the
propane is for heating in homes, businesses, and farms. In addition to keeping dwellings warm, it is
also used to cook and refrigerate foods, to dry clothes and crops, to provide
lighting, to heat water, to ripen fruits, and to fuel vehicles like tractors
and irrigation pumps. It is also used in
recreation vehicles (RV’s), and backyard grills. Like natural gas, it is a very clean-burning
fuel that emits carbon dioxide but no other harmful pollutants. In addition, propane is a raw material for
making plastic bags and other products.
Because
it is made from petroleum or natural gas, propane’s supply is limited. Currently 20-30% of the propane produced is
produced from imported oil. In many
ways, it is a superior fuel because it has as high or higher octane rating than
gasoline. It produces fewer pollutants
than regular gasoline and because of this extends the engine life. It is used less than gasoline because it is
not as easily available as gasoline, engines need to be adjusted to burn
propane, and there is a slight drop in the mileage when propane is used. If we look at the energy conversions that
occur here, we will see that they are the same as with the other fossil fuels –
radiant to chemical to heat and of course some light as in any burning process.
Uranium
Uranium is the heaviest of
the naturally occurring elements. It is
abundant all over the world but needs to be mined and processed before it can
be used as a “fuel” for nuclear power plants.
Uranium is a radioactive element, which means that it has an unstable
nucleus or one that emits particles from time to time. To make this useful for a chain reaction, the
uranium must be purified and processed.
Only one form or isotope of uranium is useful for fission,
Uranium-235. In order to concentrate
this form, the uranium is first dissolved from its ore using acid. When dried, this form is called
yellowcake. This purified uranium is
then made into a gas called uranium hexafluoride and passed through filters
with very tiny holes. Because
uranium-235 is a smaller atom than uranium-238, it will pass through the holes
leaving the other isotope behind. This
process is known as enrichment because it increases the concentration of the
needed uranium-235 isotope. Finally, at
a fabrication plant the uranium is fused with a ceramic (clay product) and made
into barrel-shaped pellets. This allows
the uranium to be able to withstand very high temperatures. These pellets are then placed into 12-foot
metal tubes that are bundled together and called fuel assemblies. These assemblies are now ready to go into the
reactor. This is where the chain
reaction of fission occurs. In addition
water is used to provide cooling, and control rods are used to absorb neutrons
to slow the reaction if needed.
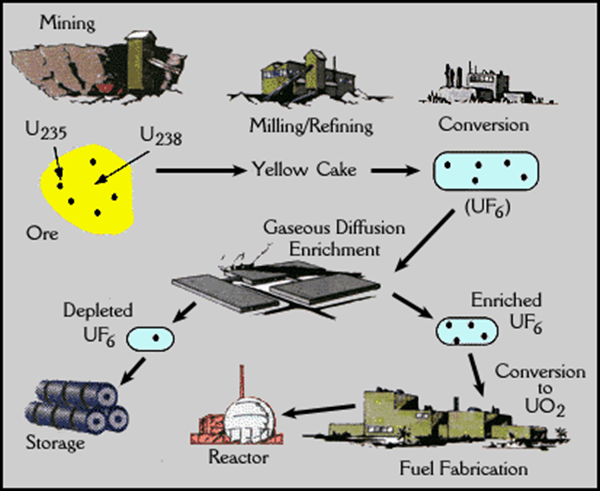
The steps in the production of uranium from mining
through enrichment to the reactor.
Fuel pellets are about the
size of your fingertip but can provide as much energy as 120 gallons of oil! How can this be? Even though uranium is called a “fuel” for
the nuclear reactor, it is not a fuel in the sense that it is burned. Instead, the nuclear reaction of fission is
occurring. This involves the splitting
of the uranium nucleus with a fast-moving neutron. When the nucleus splits, other neutrons from
this nucleus fly off to strike other uranium nuclei. Two other smaller nuclei result from each
split plus some matter is completely destroyed and converted to energy. This creates a huge amount of energy in the
form of heat and light. Remember
Einstein’s familiar equation: E = mc2
which shows us mathematically how a small amount of matter can create large
amounts of energy. The energy
transformation for uranium, therefore, is different from that for the fossil
fuels. Here we begin with nuclear energy
which is transformed into heat or thermal energy and light or radiant
energy. From then on the transformations
are the same as in any other electric power plant.
With such a huge supply of
energy available through uranium you may wonder why only 8% of our energy
supply is from this source. The answer
is that there are some drawbacks to its use.
Fortunately, there are no air pollutants since there is no actual
burning with nuclear energy. There are,
however, waste products. When the
pellets are no longer useful for the reactor because the radioactivity is too
low, they cannot be carelessly discarded because they still contain enough
radioactivity to be harmful to organisms.
This spent fuel is now being stored temporarily at the nuclear power
plants in deep pools of water. In one
year 80% of the radioactivity will be lost and in ten years about 90% of it
will be lost.
But this is to be only
temporary storage until the fuel can be reprocessed to be used again or
permanently stored. When the rods are
removed about one-third of the fuel is still good, and reprocessing removes
this fuel for suitable use. None is
being reprocessed now because it costs more for reprocessing than to make new
fuel from uranium. Permanent storage
involves placing the radioactive material where it is shielded from living
things. Such a place is called a
geologic repository. This is a site deep
underground surrounded by stable rock formations in an area that is not likely
to have earthquakes or volcanic action.
The U.S. government would like to store this waste at a site in Nevada
called Yucca Mountain. This is in a very
sparsely populated area, but so far the people of Nevada have not been in favor
of this proposal. At present, there is
no national repository for permanently storing nuclear wastes.
Still, nuclear energy is the
second most used energy source for the production of electricity in the United
States. While coal is still the number
one fuel, one-fifth of the electricity generated in the United States is from
nuclear power plants. Uranium supplies
should last at least for 500 years at the present rate of use. Uranium itself is not an expensive commodity
but the cost of construction, licensing, and inspecting nuclear plants is
high. We have 66 nuclear power plants in
operation, and no new plants are scheduled to be built. In contrast, France generates 75% of its
electricity using nuclear power. Perhaps
when we solve the problem of what to do with nuclear waste, we will increase
our reliance upon this energy source.
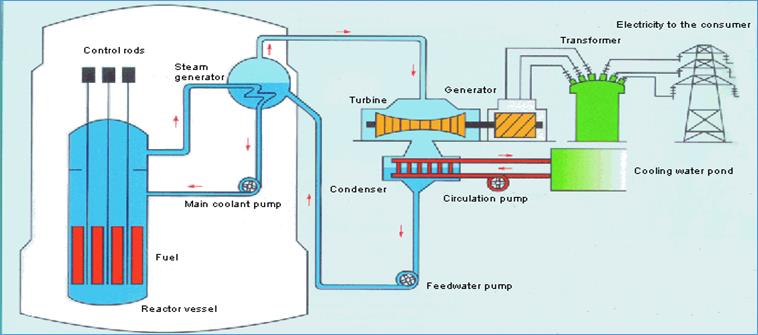
Diagram of a nuclear-powered
generating plant.
The fuel rods are shown in
red; the control rods are black. Hot
water from the reactor heats other water and converts it to steam. The steam drives a turbine which turns a
generator. It is the generator that
creates the electricity. There are a
number of energy conversions here:
nuclear to heat to mechanical to electrical energy.
Electricity
Production
Although electricity is not a
source of energy, as we stated before, it is a very important energy form used
in this country. Electricity is produced
from several sources of energy – coal, uranium, petroleum, natural gas, and
propane. All power plants are about 35%
efficient which means that 65% of the original energy in the fuel is converted
into wasted heat. Since most of these
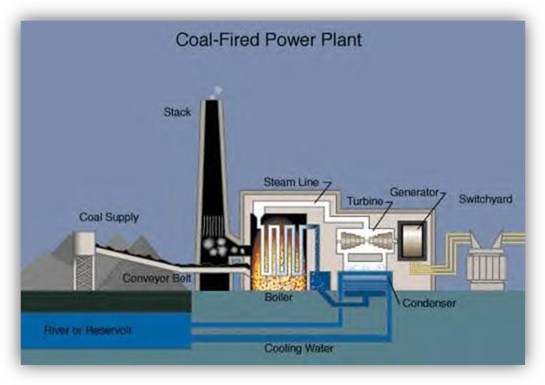
plants are coal-fired, we will study this plant in some detail. Look at the diagram on this page as each step
is described. Most often, coal is
brought to the power plant by rail or by river barge. River transportation using barges is the
least expensive. Once at the plant, it
is moved by conveyor belt into the boiler, a large furnace designed to heat
water to the boiling point and above.
Because the water is trapped in a closed area, it is under pressure and
can be heated above 100 degrees Celsius.
The super-heated steam is then taken through pipes to the turbine. The turbine is like a fancy fan with many
different sizes of blades, in series one after another. The steam causes the turbine to spin just
like wind blowing against a fan that is not turned on can cause it to
move. The moving turbine moves a part of
the generator, either the coils of wire or the magnet. In either case, an electric current is
created in the coil of wire which is then sent to homes, industries, and
businesses over wires after being transformed to the proper voltage at the
switchyard. The steam is condensed back
into water and sent back to the boiler.
The water used to cool the steam is either sent to a cooling reservoir
or a cooling tower. At the base of the
stack, electrostatic precipitator plates remove much of the fly ash. Tracing
the energy conversion in this process starts with radiant energy from the sun
to stored chemical energy in the coal to heat and light from the burning of the
coal to mechanical energy in the moving turbine and generator to electrical
energy created in the generator.
Unit Conclusion
To summarize the conversion of nonrenewable energy sources, most are fossil fuels that are burned to produce heat and light for heating buildings, cooking food, purifying metals, or for manufacturing purposes. At times, this heat is used to boil water and create steam, which creates mechanical energy in a turbine, which connects to a generator which creates electricity. The exception to this is uranium a source of nuclear energy, which produces heat through nuclear fission. This heat is used to heat water to create steam just as the fossil fuels are. The more transformations of energy that are performed during a process, the more energy that is lost to wasted heat. This is reflected in the efficiency of the process with 100% efficiency signifying no wasted energy.





