Required Materials for Science Physical
Science
Please print or save this document for future reference.
![j0197953[1]](SCIPSIU19Historical_History_image003.png)
HISTORICAL
PERSPECTIVES AND GROWTH THROUGHOUT HISTORY
Scientific studies are
typically ongoing processes. The premise of the original study may be rejected
by some scientists and supported by others. It is important to realize that
currently accepted scientific models evolve as scientists continue to study all
of the previous research. In some cases, entire theories are changed based on
newly emerging evidence. As strange as this may seem today, there was a time in
ancient history when it was thought that the world was flat. This early theory
has obviously been discarded due to evidence which proved that the earth is
round, but scientific research can often be limited by the tools, information,
and experiences available to 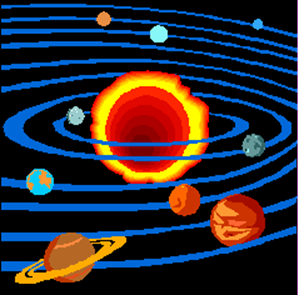 the scientist. Scientific research and technology
often aid each another. Early theories of the planets placed the earth at the
center of the solar system. It was generally accepted that humans were the most
important beings, so everything in the universe must revolve around us. Today,
we know that the sun is the center of our solar system and the planets orbit
the sun. Advances in technology, such as the telescope, have helped to prove
the current model which we now know to be true.
the scientist. Scientific research and technology
often aid each another. Early theories of the planets placed the earth at the
center of the solar system. It was generally accepted that humans were the most
important beings, so everything in the universe must revolve around us. Today,
we know that the sun is the center of our solar system and the planets orbit
the sun. Advances in technology, such as the telescope, have helped to prove
the current model which we now know to be true.
The atomic theory is another
theory well known that has undergone change over time. In most cases, entire
theories or models are not completely discarded. The theory or model is revised
as new research adds to clarity and details which were not completely
understood earlier. A scientist by the name of Dalton theorized in 1803 that an
atom was simply a solid sphere and, this theory was accepted for nearly 100
years. In 1897, however, J.J. Thomson stated that an atom was a ball of positive
electricity with negative particles embedded in it. This research was built
upon by Bohr and Rutherford in the early 1900’s and, they concluded that an
atom was made of a positively charged center orbited by electrons and these
electrons orbited the nucleus much like the planets orbit the sun. The electron
cloud model was proposed in 1926 and again changed the thinking on how
electrons orbit the nucleus.

Figure 1 John Dalton
(1766-1844)
![]() John Dalton's Particle Theory (00:37)
John Dalton's Particle Theory (00:37)

Figure 2 Ernest Rutherford
(1871-1937)
![]() Ernest Rutherford
and the Structure of the Atom (01:55)
Ernest Rutherford
and the Structure of the Atom (01:55)

Figure 3 J. J. Thompson
(1856-1940)
![]() J.J. Thompson Discovers
Electrons (00:41)
J.J. Thompson Discovers
Electrons (00:41)
![]() Niels Bohr's Atomic Model (01:24)
Niels Bohr's Atomic Model (01:24)
![]() Atomic Number, Mass Number, Isotopes (02:19)
Atomic Number, Mass Number, Isotopes (02:19)
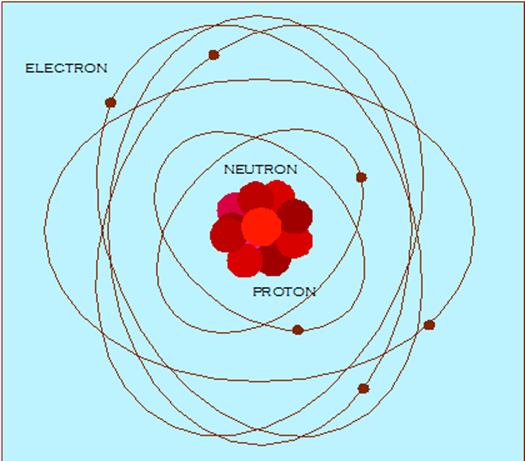
Today, continuing research is causing us to redefine
our thinking on the structure of an atom. Each of the earlier studies provided
excellent information about the atom and new studies simply improve on the
earlier models. The previous theories are not discarded, but are revised.
Scientific research has now proven that atoms are composed of parts that are
smaller than protons, neutrons, and electrons. These new subatomic particles
are continually being studied as technology and science develop better tools
with which to conduct better research. As science advances, the knowledge as to
how to create better technology emerges and we continue to see how science and
technology complement each another.
Although many of the
fundamental scientific theories may have roots dating back hundred or even
thousands of years ago, they continue to impact our lives. Newton’s laws have
been a fundamental part of our scientific knowledge for years, and are used in
the building of many modern devices. Newton’s laws influence such things as
simple as the shoes you are wearing to the highly technical space shuttle that
carry astronauts into space. The effects of scientific discoveries have had
profound influences on society throughout time with those effects being
positive as well as negative. Nuclear science has provided many great advances,
such as nuclear power which is very abundant, clean, and cost effective. The
use of nuclear bombs, however, threatens to end life on the planet as it is now
known.
 Now
answer questions 1-15.
Now
answer questions 1-15.